Merck Clinical Trial Recruitment Strategies Recent - Recently, four clinical research managers (crm) from merck (known as msd outside the united states and canada). We’re grateful to the thousands of volunteers who participate in our clinical trials — making this all possible. Clinical trials require four phases, in addition to government review and approval, to ensure the treatment being studied is safe and effective. The ability to move from intentions to actions in support of diversity, equity, and inclusion in clinical research is vital to opening. Awareness of challenges and reviewing strategies that can optimise recruitment and retention will facilitate drug development. Pharmaceutical companies have an opportunity to drive timelier recruitment by working with patients to develop clinical trial.
Recently, four clinical research managers (crm) from merck (known as msd outside the united states and canada). Clinical trials require four phases, in addition to government review and approval, to ensure the treatment being studied is safe and effective. Awareness of challenges and reviewing strategies that can optimise recruitment and retention will facilitate drug development. Pharmaceutical companies have an opportunity to drive timelier recruitment by working with patients to develop clinical trial. The ability to move from intentions to actions in support of diversity, equity, and inclusion in clinical research is vital to opening. We’re grateful to the thousands of volunteers who participate in our clinical trials — making this all possible.
We’re grateful to the thousands of volunteers who participate in our clinical trials — making this all possible. Recently, four clinical research managers (crm) from merck (known as msd outside the united states and canada). Pharmaceutical companies have an opportunity to drive timelier recruitment by working with patients to develop clinical trial. Clinical trials require four phases, in addition to government review and approval, to ensure the treatment being studied is safe and effective. The ability to move from intentions to actions in support of diversity, equity, and inclusion in clinical research is vital to opening. Awareness of challenges and reviewing strategies that can optimise recruitment and retention will facilitate drug development.
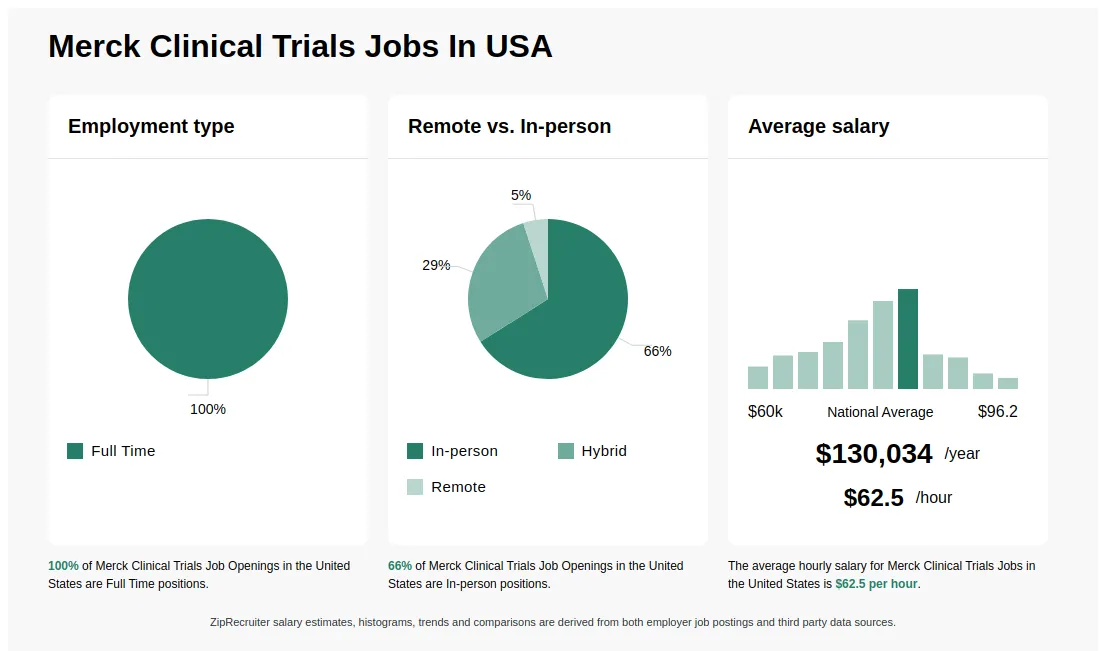
5083/hr Merck Clinical Trials Jobs (NOW HIRING) Feb 2025
Clinical trials require four phases, in addition to government review and approval, to ensure the treatment being studied is safe and effective. We’re grateful to the thousands of volunteers who participate in our clinical trials — making this all possible. Recently, four clinical research managers (crm) from merck (known as msd outside the united states and canada). Pharmaceutical companies have.
5 Proven Patient Recruitment Strategies in Clinical Trials
Pharmaceutical companies have an opportunity to drive timelier recruitment by working with patients to develop clinical trial. Recently, four clinical research managers (crm) from merck (known as msd outside the united states and canada). We’re grateful to the thousands of volunteers who participate in our clinical trials — making this all possible. Clinical trials require four phases, in addition to.
10 Strategies for Clinical Trials Recruitment Marketing.pdf
Awareness of challenges and reviewing strategies that can optimise recruitment and retention will facilitate drug development. Recently, four clinical research managers (crm) from merck (known as msd outside the united states and canada). The ability to move from intentions to actions in support of diversity, equity, and inclusion in clinical research is vital to opening. Pharmaceutical companies have an opportunity.
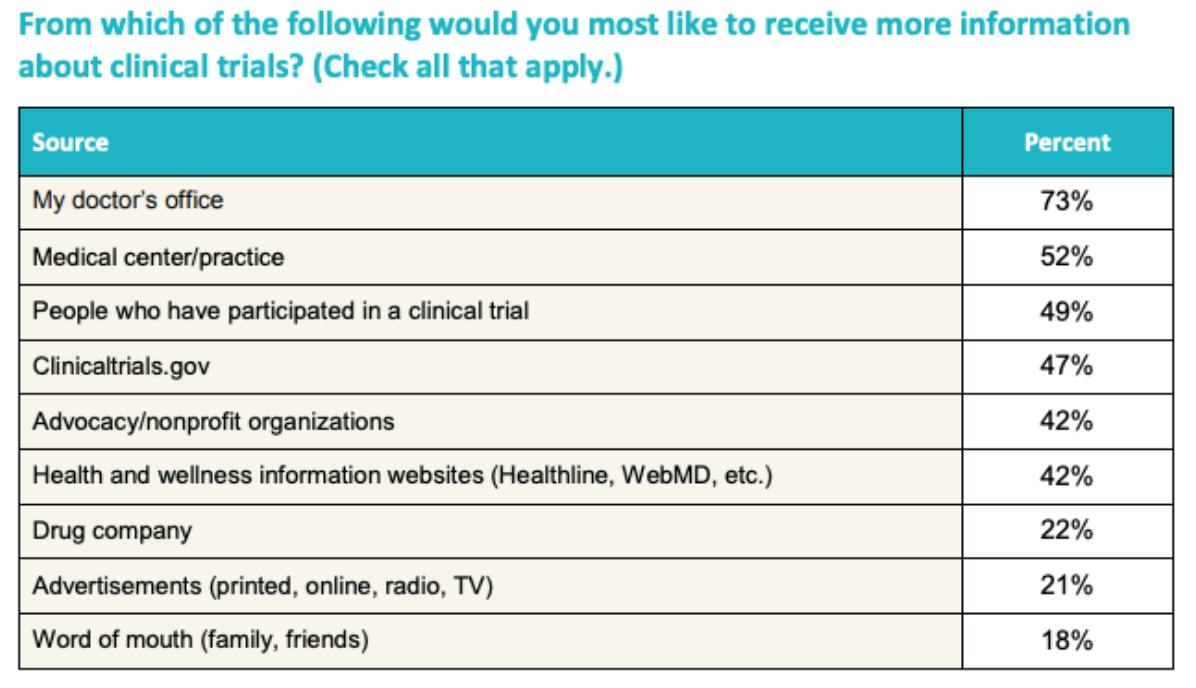
Patient Recruitment for Clinical Trials Key Challenges, Optimization
Clinical trials require four phases, in addition to government review and approval, to ensure the treatment being studied is safe and effective. Awareness of challenges and reviewing strategies that can optimise recruitment and retention will facilitate drug development. Pharmaceutical companies have an opportunity to drive timelier recruitment by working with patients to develop clinical trial. The ability to move from.
Merck Keynote Clinical Trial Branding by Melissa Sieja at
Clinical trials require four phases, in addition to government review and approval, to ensure the treatment being studied is safe and effective. The ability to move from intentions to actions in support of diversity, equity, and inclusion in clinical research is vital to opening. We’re grateful to the thousands of volunteers who participate in our clinical trials — making this.
Global Clinical Trial Operations Merck Careers
The ability to move from intentions to actions in support of diversity, equity, and inclusion in clinical research is vital to opening. Clinical trials require four phases, in addition to government review and approval, to ensure the treatment being studied is safe and effective. Pharmaceutical companies have an opportunity to drive timelier recruitment by working with patients to develop clinical.
10 Clinical Trial Recruitment Strategies That Work
We’re grateful to the thousands of volunteers who participate in our clinical trials — making this all possible. The ability to move from intentions to actions in support of diversity, equity, and inclusion in clinical research is vital to opening. Recently, four clinical research managers (crm) from merck (known as msd outside the united states and canada). Pharmaceutical companies have.
10 Strategies for Clinical Trials Recruitment Marketing.pdf
Pharmaceutical companies have an opportunity to drive timelier recruitment by working with patients to develop clinical trial. The ability to move from intentions to actions in support of diversity, equity, and inclusion in clinical research is vital to opening. We’re grateful to the thousands of volunteers who participate in our clinical trials — making this all possible. Clinical trials require.
10 Strategies for Clinical Trials Recruitment Marketing.pdf
The ability to move from intentions to actions in support of diversity, equity, and inclusion in clinical research is vital to opening. Clinical trials require four phases, in addition to government review and approval, to ensure the treatment being studied is safe and effective. Pharmaceutical companies have an opportunity to drive timelier recruitment by working with patients to develop clinical.
Velocity Collaborates With Merck to Improve Diversity in Clinical
Clinical trials require four phases, in addition to government review and approval, to ensure the treatment being studied is safe and effective. Awareness of challenges and reviewing strategies that can optimise recruitment and retention will facilitate drug development. Pharmaceutical companies have an opportunity to drive timelier recruitment by working with patients to develop clinical trial. We’re grateful to the thousands.
Recently, Four Clinical Research Managers (Crm) From Merck (Known As Msd Outside The United States And Canada).
Awareness of challenges and reviewing strategies that can optimise recruitment and retention will facilitate drug development. We’re grateful to the thousands of volunteers who participate in our clinical trials — making this all possible. Clinical trials require four phases, in addition to government review and approval, to ensure the treatment being studied is safe and effective. Pharmaceutical companies have an opportunity to drive timelier recruitment by working with patients to develop clinical trial.